|
11/20/2023 0 Comments Phase changes 
There is a TABLE of Phase Change Data in the Appendix So always double check the units that your are given - you might have to convert from moles to grams or vice versa. 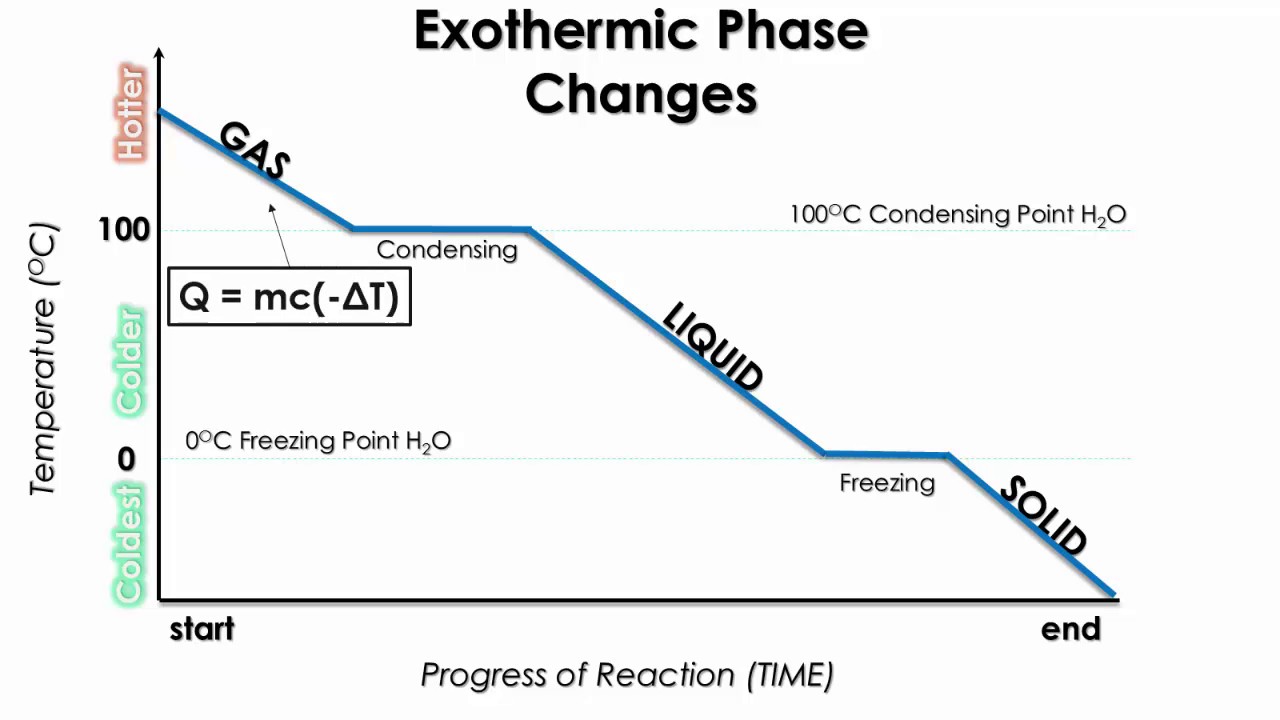
For example, if I want to calculate the amount of heat that will melt 25 grams of ice at 0 ☌ into 25 grams of water at 0 ☌, I'd do the following.Īnd to reiterate, just like with heat capacities, you can also have heats of transitions in terms of moles instead of grams. Where the "trans" is for which ever transition you are calculating for. 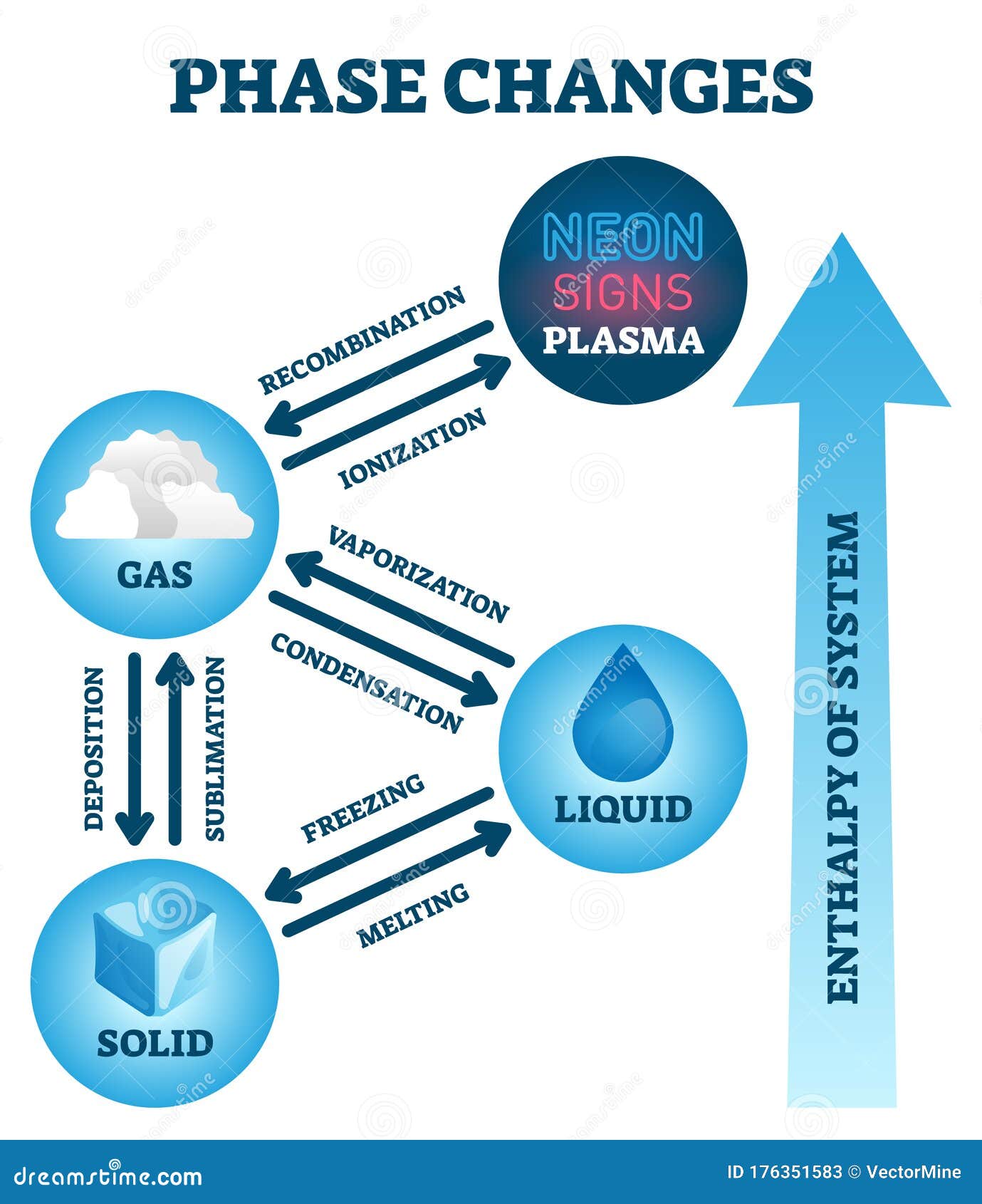
Be sure and check the units on your Δ H value to see which amount you need to use: grams or moles. The "amount" can be mass in grams or amount in moles. So the calculation of the heat required just needs the Δ H value and the amount of substance. The value (under standard conditions) of the heat of sublimation of a substance is equal to the sum of the heats of fusion and vaporization (Δ H sub = Δ H fus + Δ H vap ).ĪLL these transitions occur at the transition temperature and there is no change in temperature throughout the transition. The heat required for the transition is the heat/enthalpy of sublimation, Δ H sub. The temperature of the transition is the sublimation point (solid to gas). This is because you must overcome ALL the IMFs of the substance to transition to gas phase. notice how much bigger (almost 7×) the heat of vaporization is compared to the heat of fusion. The heat of condensation is just the negative (exothermic) of that value. Water has a heat of vaporization of 2260 J/g. The amount of heat to cause this change is the heat of vaporization or Δ H vap and it is a positive quantity (endothermic). For water, the boiling point is 100 ☌ (212 ☏). Remember, it is the same temperature either way. The temperature at the liquid/gas transition is the boiling point (bp) or the condensation point (rarely referred to BTW). Going the opposite way is freezing and the heat/enthalpy of freezing is a negative quantity (exothermic) because heat is removed or released. Melting is also known as fusion and the heat required to melt a specific amount of substance is the heat or enthalpy of fusion which is symbolized as Δ H fus and it is a positive (+) quantity - meaning you ADD heat to the solid (endothermic) to get it to melt to the liquid state. As an example, water has a freezing point of 0 ☌ (32 ☏) and ice has a melting point of 0 ☌ (32 ☏). When you say freezing point, you are inferring that the liquid is freezing into a solid. The temperature at which a solid melts to become a liquid is known as the melting point (mp) which is identical numerically to the freezing point (fp). Let's look at each change and get the lingo down as well as the sign convention. Each change occurs at a specific temperature as well. Note that each change has an associated name and that half of them are endothermic (the red arrows) and the other half are exothermic (the blue arrows). Really this is 3 changes with 2 directions of change possible in each. Each phase can directly change into the others and vice versa, therefore there are a total of 6 possible changes. There are 3 normal phases of matter that we are concerned with - solid, liquid, and gas. All bets are off on this premise when there is a phase change. As long as the substance doesn't change its physical phase this works. The input heat results in a higher temperature of the substance. The specific heat capacity is used in calculations where a given substance is heated from one temperature to another.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
